Producing ethanol from biomass with bacteria
The following post was derived from the University of Rochester (NY) website for press releases. You might ask why I think it might be of interest to you--and if you do, my answer is that ethanol is the wave of the future. Not because it's a good idea, but because the government in all its glory and wisdom is backing this energy play. Already ethanol production is having strong effects on the price of corn and it's only going to get worse--the research detailed here might change all that by allowing ethanol production to be shifted to waste biomass like sugar cane pulp and grass.
Researchers at the University of Rochester have identified how genes responsible for biomass breakdown are turned on in a microorganism that produces valuable ethanol from materials like grass and cornstalks. This is very important since most ethanol is now produced from corn which isn't very efficient and also drives up the price of corn--and any animal or food "made" from corn.
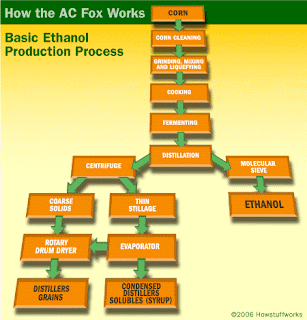 On the other hand, waste products such as grass clippings and wood chips—once thought too difficult to turn into ethanol—may soon be able to replace corn by using bioengineered bacteria to break down these cellulose based wastes.
On the other hand, waste products such as grass clippings and wood chips—once thought too difficult to turn into ethanol—may soon be able to replace corn by using bioengineered bacteria to break down these cellulose based wastes.
These findings were published in Proceedings of the National Academy of Sciences (PNAS) on Thursday and may allow scientists to engineer ethanol-producing super-organisms that can make clean-burning fuel from the nation's one billion unused tons of yearly biomass waste.
"This is the first revelation of how a bacterium chooses from its more than 100 enzymes to break down a particular biomass," says David H. Wu, professor in the Department of Chemical Engineering at the University of Rochester. "Once we know how a bacterium targets a particular type of biomass, we should be able to boost that process to draw ethanol from biomass far more efficiently that we can today."
Ethanol holds the promise of a clean, renewable alternative to fossil fuels, but deriving it from plants is difficult. Producing it from corn is the easiest method, but also involves the energy spent fertilizing, maintaining, and harvesting a crop like corn. In the end, the ethanol requires nearly as much energy to produce as you can get out of it. Conversely, deriving ethanol from plant materials such as the corn stalks and wood chips is challenging because the plants' cellulose is a very tough substance to break down, making for an inefficient process. It seems you just can't win!
Wu's technique may prove much more effective than traditional methods. Instead of using separate steps to break down biomass into glucose and ferment the glucose into ethanol, as is currently done, Wu is working on a way to make a bacterium break down and ferment plant biomass efficiently in just one step.
Wu investigated C. thermocellum, which is a microorganism that has that ability to turn biomass into ethanol in one step, but is not used at the industrial scale yet because the first step, breaking down the plant's cellulose, is much too inefficient. The key, Wu surmised, is to find out what enzymes the bacterium uses to accomplish its feat, and then boost its ability to produce those enzymes. The problem, however, lies in the fact that C. thermocellum uses more than 100 enzymes, and any of the millions of combinations of them may be the magic mixture to break down a particular biomass.
So, Wu decided to make the bacterium do the work for him.
"The bacteria know how to express just the right genes to break down any particular biomass substrate, and we wanted to know how they know to turn on and off just the right genes at the right time to do the trick," says Wu. "We found the bacterium essentially throws the whole bowl of spaghetti at the wall, sees what sticks, and then makes a lot of that particular noodle."
C. thermocelllum produces low levels of many of its enzymes at any one time. When the bacterium comes in contact with wood, for instance, a few of its enzymes break down some of that wood. A product of that tiny reaction is a sugar called laminaribiose that diffuses into the cell. There it deactivates a repressor for two genes, which wake up and start pumping out the two triggers the full production of wood-degrading enzymes CelC and LicA.
Wu's paper shows the first time the triggering pathway for enzyme production in this bacterium has been revealed, and it was only possible because C. thermocellum genome was just recently sequenced. With its 100 busy enzymes, the entire genome had to be observed as a whole, since fiddling with combinations of two, three, or more enzymes at a time would have taken "more than our lifetime," Wu says.
 The picture above illustrates the effect that C. thermocellum has on cellulose. As you can see, the bacterium dissolves the strip over time just as it would grass or sugar cane waste. You can read more about this on this page which is the location that I borrowed the picture above from.
The picture above illustrates the effect that C. thermocellum has on cellulose. As you can see, the bacterium dissolves the strip over time just as it would grass or sugar cane waste. You can read more about this on this page which is the location that I borrowed the picture above from.
Researchers at the University of Rochester have identified how genes responsible for biomass breakdown are turned on in a microorganism that produces valuable ethanol from materials like grass and cornstalks. This is very important since most ethanol is now produced from corn which isn't very efficient and also drives up the price of corn--and any animal or food "made" from corn.
 On the other hand, waste products such as grass clippings and wood chips—once thought too difficult to turn into ethanol—may soon be able to replace corn by using bioengineered bacteria to break down these cellulose based wastes.
On the other hand, waste products such as grass clippings and wood chips—once thought too difficult to turn into ethanol—may soon be able to replace corn by using bioengineered bacteria to break down these cellulose based wastes.These findings were published in Proceedings of the National Academy of Sciences (PNAS) on Thursday and may allow scientists to engineer ethanol-producing super-organisms that can make clean-burning fuel from the nation's one billion unused tons of yearly biomass waste.
"This is the first revelation of how a bacterium chooses from its more than 100 enzymes to break down a particular biomass," says David H. Wu, professor in the Department of Chemical Engineering at the University of Rochester. "Once we know how a bacterium targets a particular type of biomass, we should be able to boost that process to draw ethanol from biomass far more efficiently that we can today."
Ethanol holds the promise of a clean, renewable alternative to fossil fuels, but deriving it from plants is difficult. Producing it from corn is the easiest method, but also involves the energy spent fertilizing, maintaining, and harvesting a crop like corn. In the end, the ethanol requires nearly as much energy to produce as you can get out of it. Conversely, deriving ethanol from plant materials such as the corn stalks and wood chips is challenging because the plants' cellulose is a very tough substance to break down, making for an inefficient process. It seems you just can't win!
Wu's technique may prove much more effective than traditional methods. Instead of using separate steps to break down biomass into glucose and ferment the glucose into ethanol, as is currently done, Wu is working on a way to make a bacterium break down and ferment plant biomass efficiently in just one step.
Wu investigated C. thermocellum, which is a microorganism that has that ability to turn biomass into ethanol in one step, but is not used at the industrial scale yet because the first step, breaking down the plant's cellulose, is much too inefficient. The key, Wu surmised, is to find out what enzymes the bacterium uses to accomplish its feat, and then boost its ability to produce those enzymes. The problem, however, lies in the fact that C. thermocellum uses more than 100 enzymes, and any of the millions of combinations of them may be the magic mixture to break down a particular biomass.
So, Wu decided to make the bacterium do the work for him.
"The bacteria know how to express just the right genes to break down any particular biomass substrate, and we wanted to know how they know to turn on and off just the right genes at the right time to do the trick," says Wu. "We found the bacterium essentially throws the whole bowl of spaghetti at the wall, sees what sticks, and then makes a lot of that particular noodle."
C. thermocelllum produces low levels of many of its enzymes at any one time. When the bacterium comes in contact with wood, for instance, a few of its enzymes break down some of that wood. A product of that tiny reaction is a sugar called laminaribiose that diffuses into the cell. There it deactivates a repressor for two genes, which wake up and start pumping out the two triggers the full production of wood-degrading enzymes CelC and LicA.
Wu's paper shows the first time the triggering pathway for enzyme production in this bacterium has been revealed, and it was only possible because C. thermocellum genome was just recently sequenced. With its 100 busy enzymes, the entire genome had to be observed as a whole, since fiddling with combinations of two, three, or more enzymes at a time would have taken "more than our lifetime," Wu says.
 The picture above illustrates the effect that C. thermocellum has on cellulose. As you can see, the bacterium dissolves the strip over time just as it would grass or sugar cane waste. You can read more about this on this page which is the location that I borrowed the picture above from.
The picture above illustrates the effect that C. thermocellum has on cellulose. As you can see, the bacterium dissolves the strip over time just as it would grass or sugar cane waste. You can read more about this on this page which is the location that I borrowed the picture above from.
Comments
michele sent me over today to get all edu-macated. :)
Thanks for dropping by my blog in the week - your kind comment was much appreciated as I was having a baaaaad time. Just been catching up on the blogs I usually visit including yours!
I feel so much more enlightened during the second read (I came by earlier and read this post) -- It's interesting how enzymes deal with so many products.
Thanks for educating us!
Michele sent me by, but I always come by anyhow!
Michele sent me again.
Michele sent me to say hello this time!